Isothermal titration calorimeter
Instrument Specification:

Instrument Specification:
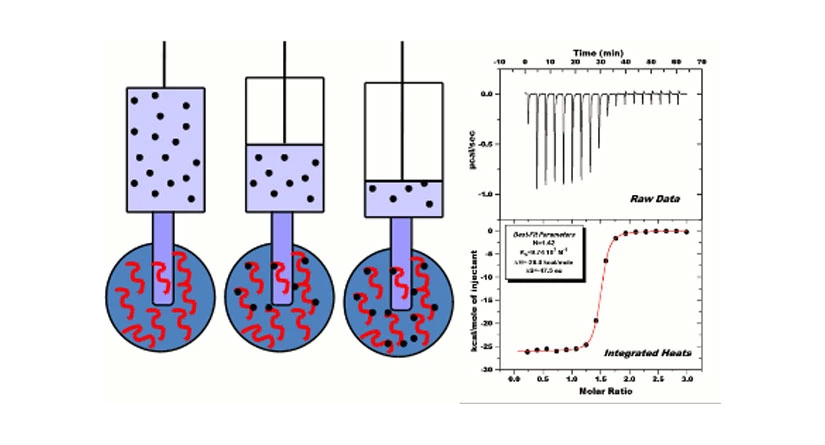
Isothermal Titration Calorimetry (ITC) is the gold standard for measuring biomolecular interactions . ITC 200 simultaneously determines all binding parameters (n, K, ΔH and ΔS) in a single experiment-information that cannot be obtained from any other method. When substances bind, heat is either generated or absorbed . ITC is a thermodynamic technique that directly measures the heat released or absorbed during a biomolecular binding event. Measurement of this heat allows accurate determination of binding constants (KB), reaction stoichiometry (n), enthalpy (ΔH) and entropy (ΔS), thereby providing a complete thermodynamic profile of the molecular interaction in a single experiment.Because ITC goes beyond binding affinities and can elucidate the mechanism of the molecular interaction, it has become the method of choice for characterizing biomolecular interactions.
How does it work?
ITC systems use a cell feedback network (CFB) to differentially measure and compensate for heat produced or absorbed between the sample and reference cell. Twin coin-shaped cells are mounted in a cylindrical adiabatic environment, and connect to the outside through narrow access tubes. A thermoelectric device measures the temperature difference between the two cells and a second device measures the temperature difference between the cells and the jacket. As chemical reactions occur in the sample cell, heat is generated or absorbed. The temperature difference between the sample and reference cells (ΔT1) is kept at a constant value (i.e. baseline) by the addition or removal of heat to the sample cell, as appropriate, using the CFB system. The integral of the power required to maintain ΔT1 = constant over time is a measure of total heat resulting from the process being studied.

Mass Spectrometers
Matrix Assisted Laser Desorption Time of Flight Mass Spectrometer(MALDI TOF

Desorption Electrospray Ionization
Instrument Specification: The LTQ XL is a high resolution linear

Gas Chromatograph Mass Spectrometer
Instrument Specification: The GCMS (Gas Chromatograph Mass Spectrometer) instrument is

Balzers Thermostar
Instrument Specification: Mass range : 1-300 amuResolution : 0.1Capillary :

SYNAPT G2-Si High Definition MS (HDMS) System
Travelling Wave Ion Mobility Mass Spectrometer: SYNAPT G2-Si High Definition

Inductively Coupled Plasma Mass Spectrometer
Instrument Specification: Instrument: NexION 300X – Perkin Elmer Plasma source: